Imaging method allows for unprecedented view of developing mouse placenta in real time
In pregnancy, the placenta is a transient yet crucial organ, supplying the fetus with oxygen and nutrients essential for survival. While its function is critical, there is still much about the placenta that we do not fully understand, including the intricacies of its development. Yet its deep anatomical location, its ability to move, and the delicate nature of pregnancy have limited researchers’ ability to obtain detailed images of the placenta in real time—until now.
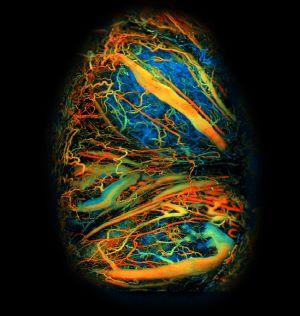
A multidisciplinary group of NIH-funded scientists have successfully captured real-time, high-resolution images of the developing mouse placenta during the course of pregnancy. Their technique, which combines a surgically implanted window with a next-generation imaging system, provides key insight into placental development under both healthy and pathological conditions, including a model of preeclampsia and alcohol consumption. The study was published today on the cover of Science Advances.
“Our real-time, live animal imaging method allows for the direct observation of the placenta’s development and response to various stressors throughout pregnancy,” said senior study author Junjie Yao, Ph.D., associate professor of biomedical engineering at Duke University. “The imaging platform’s ability to capture the intricacies of placental blood flow, oxygenation, molecular transportation, and structural changes in this animal model could advance our understanding of placental biology and its impact on pregnancy outcomes.”
A window of opportunity
Implantable windows have been previously used to capture images of tissues inside the body of animals, such as the brain and organs in the abdominal cavity. But generating images of the placenta through a window is difficult for multiple reasons, explained Yao. First, the placenta is a delicate and temporary organ that is highly sensitive to stress, and any perturbations must be taken with the utmost care. Second, the placenta is extremely dynamic, with its dramatic changes in size during development and its constant movement.
“Developing a placental window for imaging has been exceptionally challenging, because the organ is constantly moving around—the breathing of the animal is one factor, but the fetus and the placenta itself are moving as well, resulting in enormous motion artifacts,” said Yao. “We’ve been working on real-time imaging of the placenta for a long time, perfecting not only the surgical installation of the window, but also optimizing the imaging technology to be as fast and as high-resolution as possible.”
To reduce image distortions due to movement and to provide stability, the researchers devised a precise surgical procedure to implant the placental window. After making an incision and exposing the placentas of the pregnant mice, the placenta is carefully sutured to the surrounding abdominal muscles to minimize its movement. A biocompatible window is then placed over the placenta and secured to the abdominal muscles. This window contains a transparent membrane that allows both light and sound to travel through, allowing for multiple types of imaging.
To provide real-time images of the highest speed, the researchers tweaked their existing photoacoustic microscopy (PAM) system to make it even faster with a larger field of view. Their ultra-fast PAM technique can image placenta vasculature and blood oxygenation levels by flashing quick light pulses that generate detectable ultrasound waves after they interact with the tissues through the window. This method allowed the researchers to track placental development daily throughout the course of pregnancy.
An unprecedented look at the placenta
PAM techniques have been previously used to track the flow of blood. This method works by pulsing specific wavelengths of light at target tissues, which absorb that energy and produce ultrasound waves which can be detected by a transducer. But traditional PAM techniques are limited by how quickly the light and sound can be steered, requiring roughly 20-30 minutes to capture a static image from the resultant ultrasound waves.
To improve this technique, Yao and colleagues developed an ultra-fast system that uses a spinning polygon scanner to rapidly direct laser pulses at target tissues, substantially decreasing the amount of time required to generate an image. In this study, the researchers made further modifications to their ultra-fast PAM technique, providing the platform with added stability and allowing the polygon scanner to spin faster still, with the ability to capture images in a fraction of a second. These state-of-the-art PAM systems allowed them to monitor the mouse placenta in real time.
The researchers first wanted to watch how the placenta developed during the course of a healthy mouse pregnancy, which lasts about 20 days. They began monitoring the placenta seven days after embryonic development and captured the first imaging evidence of a hypoxic (low oxygen) environment in the early stages of pregnancy. “This initial hypoxic condition is crucial for the placenta, as it protects the fetus from oxidative stress and triggers the rapid development of new blood vessels,” Yao explained. “Using our imaging technique, we can see that a very complex microvessel network is created early in the pregnancy to facilitate the later delivery of oxygen and nutrients to the fetus—it was fascinating to observe.”
The blood oxygenation levels increased dramatically in the second trimester, and then decreased again before the animals delivered. “The initial requirement for a hypoxic environment, followed by precise changes in blood flow and oxygenation, showcases the placenta's sensitivity to internal and external conditions and the intricate balance of environmental conditions needed for optimal fetal development,” said Yao.
Pathological placental conditions
After establishing a baseline, the researchers next wanted to observe placental development during pathological conditions, such as preeclampsia. Preeclampsia is a life-threatening pregnancy complication characterized by a sudden onset of high blood pressure, among other symptoms, that can ultimately lead to maternal and fetal mortality if not properly managed.
To simulate preeclampsia, the researchers injected pregnant mice with lipopolysaccharide (LPS), an inflammatory agent that induces a preeclampsia-like phenotype. Compared with untreated pregnant mice, LPS-treated mice had changes in both oxygenation levels and blood vessel development in the placenta during the early stages of pregnancy. “Instead of developing a network of tiny blood vessels, the placentas from the LPS-treated mice developed abnormal, larger blood vessels, and the environment was substantially less hypoxic,” Yao noted. “We hypothesize that the placentas from LPS-treated mice didn’t have the hypoxic trigger to develop microvessels as they should, suggesting that preeclampsia pathology may begin at an early stage of the pregnancy. Additional research in this area is clearly warranted.”
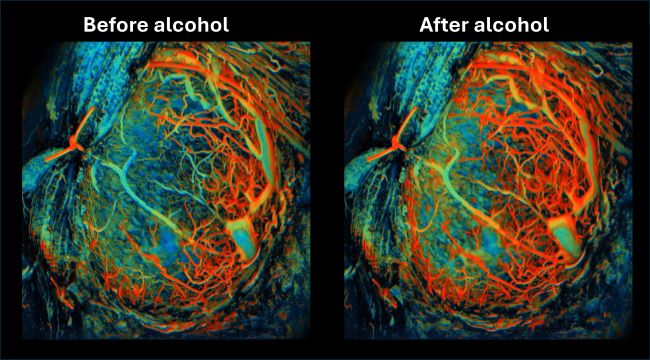
The researchers also evaluated how alcohol consumption can affect the placenta. They administered ethanol into the abdominal cavity of pregnant mice in the second trimester and observed the blood flow dynamics over a 10-minute time frame. The results were striking: the blood oxygen levels increased within the placenta by almost 30%, lasting for nearly three minutes. “This dramatic influx in oxygenation alters the delicate chemical environment surrounding the fetus, and could also jeopardize the development of microvessels within the placenta,” said Yao. “These results underscore the sensitivity of the placenta to external stressors and demonstrate how alcohol consumption can have immediate and significant effects on the placental environment.”
Lessons learned and limitations
While these images provide key insight into placental development, there are some limitations. A major limitation, beyond using a mouse to model human pregnancy, is that pups grown under the placental window were roughly 30% lighter in weight compared to their littermates that were not grown under the window. “This reduction in embryonic weight is potentially due to alterations in the placental microenvironment caused by the window implantation, such as heat loss and mechanical stress,” Yao explained. “Nevertheless, the embryos under the window were all delivered normally and developed to the same size and weight as their littermates one month post-delivery.” Further, nearly 35% of embryos grown under the window were lost, mostly within 24 hours following window implantation. According to the publication, the normal rate of spontaneous fetal resorption (pregnancy loss) in mice is 10-20%.
Yao noted that this imaging technique could be adapted for mammals with longer gestational periods than mice, such as rats and rabbits, and that future work may focus on how environmental factors, such as climate change and water pollution, could impact placental health and outcomes.
“This state-of-the-art photoacoustic imaging platform, built on years of novel iterations, opens the door to a wide range of fundamental biological discoveries,” said Randy King, Ph.D., a program director in the Division of Applied Science & Technology at NIBIB. “The technology, while impressive in its own right, is ultimately exciting because it could lead to new insights into placental development and health that could someday benefit humans.”
This study was supported by grants from NIBIB (R01EB028143), the NIH BRAIN Initiative (RF1NS115581), the National Institute of Neurological Disorders and Stroke (NINDS; R01NS111039), the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK; R01DK132889 and R01DK119795), the National Institute of General Medical Sciences (NIGMS; R35GM122465), and the National Science Foundation.
This science highlight describes a basic research finding. Basic research increases our understanding of human behavior and biology, which is foundational to advancing new and better ways to prevent, diagnose, and treat disease. Science is an unpredictable and incremental process—each research advance builds on past discoveries, often in unexpected ways. Most clinical advances would not be possible without the knowledge of fundamental basic research.
Study reference: Xiaoyi Zhu et al. Longitudinal intravital imaging of mouse placenta. Sci Adv. 10, eadk12789(2024). DOI:10.1126/sciadv.adk1278
